编者按:
肥胖作为一种慢性代谢性疾病,从多方面影响着人们的健康,还可以导致多种其它代谢性疾病的发展。目前由于人们生活、饮食习惯的变化,在当代,肥胖患者越来越多。
发现益生菌或对人体健康有一些作用,那么它对于肥胖的预防或者治疗有没有什么帮助呢?
我们特别编译了发表在 Microorganisms 上关于益生菌与肥胖的综述,以详细解答这个问题,阐述益生菌-肠道-肥胖三者之间的关系。
本文将分为上下 2 篇,今天为上半部分,以下是上半部分的编译:
摘要:
肥胖及由肥胖所引起的健康风险在现代社会中越来越受到重视。引起肥胖的主要因素有:过量的食物摄取、久坐的生活习惯、高糖和高脂的饮食习惯及遗传因素等。
本综述汇总了肠道微生物用于干预宿主代谢的最新进展,评估了肠道微生物影响宿主代谢的一些重要的分子机制,比如胆盐代谢、短链脂肪酸代谢和代谢性内毒素血症。
之前的一些研究发现了微生物与包括肥胖在内的几种疾病之间的联系。因此,本综述聚焦于肠道微生物和肥胖间的相互影响以及其影响肥胖的机制机理。
特别的,本综述重点在于介绍一些饮食代替的治疗方式,包括饮食变化和增加益生菌辅食。此外,有关粪菌移植、合生元和代谢组学等方法也在本文中有所涉及。(这在我们的下篇文章会有所详述)
关键词:肥胖;肠道微生物;益生菌;机制;饮食
正文:
简介:肥胖与微生物

肥胖是重要的健康问题之一,与许多慢性代谢疾病有相关性,比如高血压、糖尿病和血脂异常。据 2016 年的统计显示,在全球有超过 19 亿成年人面临肥胖问题1。
根据世界健康组织(World Health Organization,WHO)规定,成年人的体重指数(body mass index, BMI)大于 25 时为正常状态,当 BMI 大于 30 时可被认定为肥胖1。肥胖人群的寿命要比正常人群短 7 年2。
在环境和基因的潜在影响下,肥胖现象近年来尤为严重。导致肥胖的全球环境因素包括运动的减少、暴饮暴食、过多摄入高热量食物、食用腌制食物和某些药物的使用等。
此外,肥胖还与一些泌尿科疾病相关,比如勃起功能障碍和前列腺增生,从而对健康产生不利影响2。
粮食及农业组织(Food and Agriculture Organization, FAO)和 WTO 将益生菌(probiotics)定义为:活的微生物,当适当摄入时,可以给宿主带来健康方面的好处4。
而每个人体内都含有 1 到 2 千克的微生物,包含大约至少 1000 种不同的物种,10 万亿个微生物5,这些微生物对肠道黏膜和宿主免疫系统很重要。大部分的肠道菌群是细菌,它们占了粪便干重的 60%;真菌和原生动物也是肠道菌群的一部分,但是它们的功能尚不清楚6。
在食品产业中,有一些具有调节免疫应答的益生菌被广泛应用,比如:干酪乳杆菌(L. casei)、约氏乳杆菌(L. johnsonii)、乳酸双歧杆菌(B. lactis)、鼠李糖乳杆菌(L. rhamnosus)、酿酒酵母(S. cerevisiae)和动物双歧杆菌(B. animals)7,8。

研究发现,格氏乳杆菌 BNR17(Lactobacillus gasseri-BNR17)具有抑制脂联素和瘦素分泌的作用,能够减少脂肪组织9。
其它益生菌,比如长双歧杆菌(B.longum)、嗜酸乳杆菌(L.acidophilu s)、乳酸片球菌(P. acidilactici)和干酪乳杆菌(L. casei)等具有降胆固醇的作用10。
干酪乳杆菌可以通过抗氧化、免疫调节和抗糖尿病等作用来降低促炎因子的浓度,进而减少氧化胁迫,并抑制 CD4+T 细胞的产生11,12。(编者注:氧化胁迫是指生物体中产生了大量氧化物质如超氧基、过氧基等对生物不利的现象)
之前关于治疗肥胖的研究已经讨论了转化医学的可能。根据 Larsen 等人的观点,肥胖虽然不是一种疾病,但却是一种缺陷13。尽管之前的研究中没有提供明确的治疗方法,但是研究发现这一“缺陷”或可通过肠道微生物和代谢情况之间的联系来调整14。
因此,最近的研究聚焦于通过摄取益生菌来调控有益菌群。我们期望通过这些益生菌以调控肥胖14。
在自身免疫疾病方面,比如 I 型糖尿病,发现了与肥胖类似的进展7,15-17。
研究发现微生物群体的改变会引起机会致病菌的侵染,这些致病菌具有较强的氧化胁迫耐受能力、降低硫酸盐浓度的能力和阻碍细菌产生丁酸的能力18。
在这种情况下,某些酶会参与到葡萄糖的动态稳定过程中。当外周胰岛素产生的抗性或 B 细胞胰岛素分泌不足的情况下就会导致失调19。
Hamad 等人发现格氏乳杆菌 SBT2055 可以降低瘦型 Zucker 鼠(常见基因性肥胖的动物模型)的肠系膜组织质量和血清瘦素水平,并减小脂肪细胞大小20。
同时在肥胖或者消瘦型的 Zucker 大鼠中观察到了血清和肝脏胆固醇含量的降低、粪便脂肪酸的改善和中性固醇的排泄现象。实验所观察到的三酰基甘油、磷脂等变化是由于淋巴吸收的减少。
另外,在使用了益生菌药物后,发现总胆固醇、三酰甘油、高密度脂蛋白和低密度脂蛋白胆固醇的含量并没有发生变化21,22。然而,由于对葡萄糖的敏感性增加,血糖水平出现降低23。
类似的,研究发现养乐多代田菌(L. casei Shirota)菌株也可以使饮食诱导的肥胖小鼠对胰岛素的敏感性增加,并降低对葡萄糖的耐受性。
来源于小麦和大麦花的多种乳杆菌的发酵产物能够抑制高脂饮食动物的体重增加,减少肾前和附睾脂肪含量和血清总胆固醇含量24;补充多种双歧杆菌菌株可以抑制大鼠体重增加、血糖含量和瘦素浓度,但不引起大鼠脂肪垫的显著改变15,25。
表 1 和表 2 汇总了一系列益生菌调控肥胖的试验信息22

表1 益生菌菌株对肥胖动物模型的有效性28

表2. 益生菌与肥胖人群实验28
另外,研究发现,肥胖与硬壁菌门的增加和拟杆菌门的减少有一定联系24,26。具体地,肥胖患者的拟杆菌门显著降低,硬壁菌门的相对含量上升,这一现象或与血浆葡萄糖浓度相关27。
目前也有一些研究侧重于探究一些益生元的作用,如阿拉伯木聚糖和阿拉伯木聚糖低聚果糖。这些益生元或能调控与肥胖相关的代谢异常。本综述汇总了以往的研究,以支持益生菌或具有调控肥胖的作用。
肠道微生物的失调
肠道微生物与人类健康的联系越来越频繁且紧密。不稳定的肠道微生物也就是肠道微生物失衡会导致潜在的炎症性肠炎、艰难梭状芽胞杆菌感染、肥胖和自身免疫紊乱23,41,42。
已经有大量的研究探讨了肥胖患者的肠道微生物菌群失调现象43,44。在人体重要的共生菌中除了拟杆菌门和硬壁菌门与肥胖相关,还有许多其它微生物也与肥胖相关。在科水平上,肠杆菌科、普雷沃氏菌科均与肥胖相关。另外还发现克里斯氏菌科在低 BMI 人群中含量较高41,45。
研究发现,发生了体重降低的成年人群中乳杆菌属含量升高,但在超重或肥胖儿童中其含量也偏高46-49。拟杆菌属的含量在肥胖人群中占比较高,并且此菌的丰度与 BMI 呈正相关49。罗斯氏菌(Roseburia) 属的菌株对超重人群有一定的益处,由于肥胖人群摄入过多不易消化的多糖时,该类菌可以促进排泄50-52。
此外,研究中也发现了 BMI 和双歧杆菌之间的逆相关34。欧文氏菌属(Erwinia),琥珀酸弧菌属(Succinivibrio)53,另枝菌属(Alistipes)54和颤螺菌属(Oscillospira)55 的丰度在正常体重人群中比肥胖人群中高 56-60。
灵巧粪球菌(Coprococcus catus),Blautia hydrogenotrophica,布氏瘤胃球菌(Ruminococcus bromii),卵瘤胃球菌(Ruminococcus obeum)和凸腹真杆菌(Eubacterium ventriosum)与日本人群的肥胖现象有显著的相关性60-62。
而多形拟杆菌(Bacteroides thetaiotaomicron),Bacteroides faecichinchillae,Flavonifractor plautii,Blautia wexlerae 和 鲍氏梭菌(Clostridium bolteae)这些细菌则与体重较轻的人群相关63。
此外,肠道共生菌(Akkermansia muciniphila)已经应用于肥胖的辅助治疗中63。
这些菌株在种的水平和菌株的水平上的多样性导致了重量增加程度和肥胖程度差异性。因此,对肠道微生物和其背后的关联机制现在已经吸引了众多研究者的关注64,65 。
肥胖代谢的机制研究
一些导致肥胖的肠道菌群的功能机制如图 1 所示:
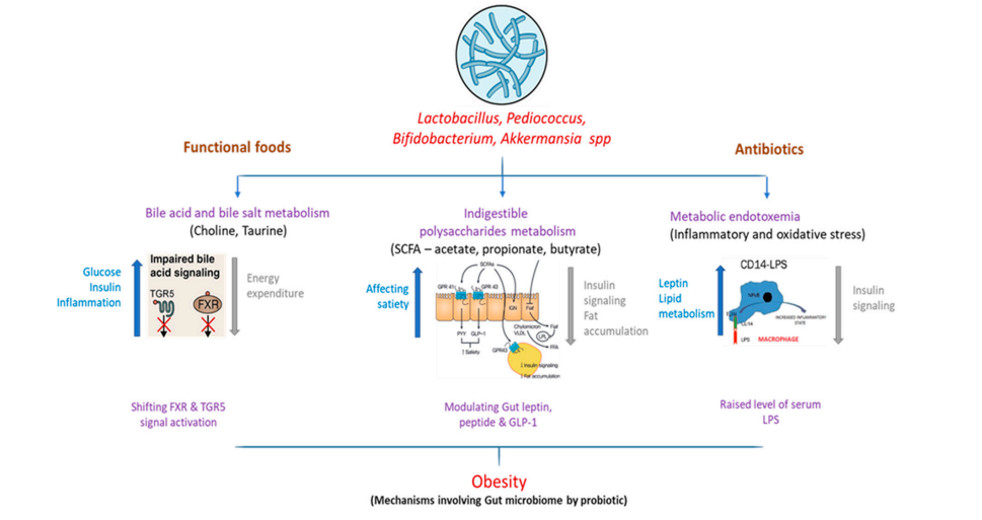
图1 肠道微生物组与下游代谢的互作对肥胖的影响。
胆汁酸与小肠中膳食脂肪的摄入息息相关。甘氨酸或者牛磺酸与在肝脏中合成的胆固醇相结合。在肠道中,细菌能够通过分解并二羟基化的方式将原胆汁酸转化为次级胆汁酸66。除了胃肠道,胆盐也可以通过破坏膜的可靠性来抑制多种肠道菌群的生长,比如一些益生菌双歧杆菌和乳杆菌67,68。
胆汁酸同样也是法尼醇 X 受体(farnesoid X-receiving receptor,FXR)的配体。在一篇有关 FXR 失活小鼠和野生小鼠的肠道微生物研究中,发现肠道微生物可以通过 FXR 信号促进饮食肥胖69。
也有研究报道,G 蛋白偶联胆汁酸受体 1(GPBAR1)/武田 G 蛋白偶联受体 5(TGR5),这些受体的活性完全由胆汁酸激活,并可以通过释放胃胰高血糖素样肽-1 来实现葡萄糖的稳态70。后续的研究也报道了 TGR5 在减肥手术中的抗肥胖功能。
高脂饮食的 FXR 失活小鼠通过粪菌移植后,发现了无菌小鼠的体重增长要比非无菌小鼠体重增长慢。这表明了肠道微生物可以引发饮食性肥胖71,72。
在远端肠道的无氧环境中,不能被胃肠道消化吸收的多糖是细菌的分解底物73。短链脂肪酸(Short-chainfatty acids,SCFAs)中的主要成分是乙酸、丙酸、丁酸 SCFAs 在人体内一天释放大约 80-200 卡路里,大多数被各种器官所消耗74。
在肥胖患者中,粪便菌群中产丁酸盐的细菌数量减少,这与饮食碳水化合物(如多糖、抗性淀粉和蔬菜低聚果糖)摄取的减少相关75 。在低纤维饮食的患者中也观察到了粪便丁酸含量、总 SCFAs 含量和双歧杆菌量的减少76。
低纤维饮食已被证明可以通过维持胃粘膜菌群的稳定性提高对病原体的敏感性。微生物所产生的 SCFAs 可以降低胆囊的 pH 值,为产生丁酸的细菌提供生态位,增加这些细菌的丰度从而改变肠道微生物组。
此外,SCFAs 除了具有提供能量和调控 pH 的功能外,SCFAs 还至少是 2 种 G 蛋白偶联受体(Gpr41、Gpr)的信号分子77。因此,高纤维饮食可以通过 SCFAs 调控胃肠道及一些代谢途径来协助肥胖的调控。
长期以来,对肥胖都通过慢性炎症和胰岛素抗性来定义的78。在一些研究报道中,肥胖可以导致肠道渗透性和代谢性内毒素血症。肥胖和慢性炎症之间的关系直到 Cani 等的研究才被阐明。
Cani 等发现高脂饮食会导致血浆脂多糖(LPS,LPS也被称为细菌内毒素)的升高,或者称之为代谢性内毒素血症79。Cani 等将代谢性内毒素血症定义为一种慢性的高血浆 LPS 紊乱症,其发病率比 LPS 败血症低 10 到 50 倍。
在正常饮食的肥胖小鼠中观察到了代谢性内毒素血症(包括促炎症和氧胁迫)。这些小鼠的正常饮食是由磨碎的小麦、玉米或燕麦、苜蓿和豆粕、含有矿物质和维生素的蛋白质组成的,因此这可能是肥胖饮食对正常小鼠诱导的结果56。

在低脂饮食(low-fat diet,LFD)的小鼠中,硬壁菌门的数量显著低于高脂饮食(high-fat diet,HFD)的小鼠。但是变形菌门的数量并没有发生显著变化80。在这些研究中,饮食内毒素的增加与脂肪沉积、全身和组织特异性炎症,以及胰岛素抗性等增强相关79,81。
在健康人群中,代谢性内毒素血症可以减少 35%的胰岛素敏感性,这与能量消耗相关,并且可能是高脂、高卡路里饮食摄入而导致的一种结果81。
横向、纵向研究还有一些实验的补充结果都强调了代谢性内毒素血症在炎性肥胖和心脏代谢异常中的临床意义。
在近几十年中,普遍认为通过饮食摄取的脂类物质可以调控炎症、先天免疫机制、代谢通路82。膳食脂类物质具有一定的营养价值,但是这些物质也在核受体的相互作用(促炎症)中发挥作用83。
这些受体包括过氧化物酶体增殖物活性受体(peroxisome proliferator active receptors,PPARs)家族和肝脏 X 受体(liver X receptors,LXRs)都是炎症代谢通路中的中枢物质。
许多脂肪酸可以激活 PPAR 家族的 3 个成员,从而增强胰岛素的作用,并抑制了促炎细胞因子如 TNF-α 的生成84,85。
所有饮食代谢物和脂类物质均可以激活 G-蛋白受体(Gprs)。比如,饮食代谢产生的乙酸可以激活 Gpr43 和削弱脂肪细胞中的脂减作用,这个现象是降低血浆游离脂肪酸水平的关键。这一结果表明了 Gpr43 在调控脂类物质代谢中的潜在作用86,87。
Backhed 等发现微生物可以增加 Fiaf 的表达和活性从而降低体重88。还有研究证明了动脉粥样硬化的病理生理与人群肠道微生物组的变化相关89。
富含磷脂酰胆碱的食物如:鸡蛋,牛奶,鱼,肝脏,家禽和红肉,是饮食中胆碱的重要来源90。胆汁酸是脂质代谢、胆固醇代谢的重要调节物质,它可以促进营养物质和微生物在肠道的吸收和转运。根据肠肝循环,当胆汁在肝脏中产生时 95%的胆汁酸被回肠末端吸收,然后被肝脏重新吸收。因此脂质分解代谢会消耗酸。
肠道微生物通过对胆汁酸的二羟基化作用将原始胆盐转化为次级胆盐66,91。当摄入低剂量的抗生素时,会影响肠道微生物的组成并影响人体胆汁酸代谢,也会对一些糖类代谢造成影响92,93 。
在鼠模型中胆汁酸可以促进 GLP-1 的分泌,导致 TGR5 受体的激活。然而,一些研究尚未找到可在减肥手术前几天使用的,针对代谢的高浓度血浆胆汁酸94。
欲望、食物摄取和能量平衡是复杂神经内分泌因子和受体网络的重要组成部分,该网络调控了胃肠道和大脑之间的代谢通路95。进食后,营养物质在消化道的移动可以激活复杂的神经和激素信号。因此,肠道菌群可以认为是第二个大脑。
越来越多的证据表明多巴胺、肾上腺素、去甲肾上腺素、γ-氨基丁酸、血清素、吲哚代谢物和其他一些肠道微生物受体可以影响饮食偏好。
由自主神经系统的传入神经纤维(如迷走神经)介导的信号,将信息从肠道发送至孤核(NTS)和效应纤维,然后投射至肠的平滑肌。NTS 信息发布至下丘脑,可以调控 ARC 神经元的能量平衡食欲和饮食摄入。ARC 提供与agouti和厌食症肽相关的致病性神经 Y 家族肽,并调节原黑皮质素的转录12,88。
其它外周胃肠道的多肽包括胰多肽和酪酪肽可以降低食欲。Neuromedin U 和 Neuromedin S 两种神经肽可以抑制食欲,而黑色素浓缩激素和两种食欲肽(orexin A 和 orexin B)则会促进食欲。
此外,Y 家族神经肽主要由肠内分泌细胞产生和分泌,而这些内分泌细胞主要聚集在胃肠道壁中96,97。
由肠道微生物产生的短链脂肪酸等代谢产物可以与肠内分泌细胞受体相结合并改善肠内激素的释放进入全身循环。
在动物和人类的研究中发现,肠道微生物发酵不可消化碳水化合物可以增加 SCFAs 的产生和肠道激素的分泌98。由肠道微生物产生的乙酸是 SCFA 的主要物质99。然而,由于微生物组的改变所导致的乙酸含量的增加可以引起副交感神经系统的激活,促进葡萄糖的转运、胃泌素的分泌和肥胖97,100。
其他对于无菌小鼠的研究发现肠甜味信号蛋白 T1R2 的上调可以导致碳水化合物的过量摄入101。某些乳酸菌可以将谷氨酸转化为 γ 氨基丁酸,并表达 GABA 结合蛋白102,103和血清素95,97,104-106,以调节食欲。
血清素可以通过调节食欲抑制作用来介导黑皮质素对食欲的影响102。在另一项对无菌小鼠的研究中,肠道甜味信号蛋白可以引起甜味营养物质的更多摄入64,68,103,107。控制体重稳态的黑素皮质素神经元的调节具有降低食欲的作用108,109。
γ 氨基丁酸作为中枢神经系统的主要抑制剂,可以促进进食和下丘脑 ARC 表达蛋白的模糊神经元的突触放电,并控制能量平衡(图 2)108,110-112。因此,阐明脑肠轴和人体微生物组之间的联系机制,可以为肥胖和代谢紊乱提供潜在的治疗方法。
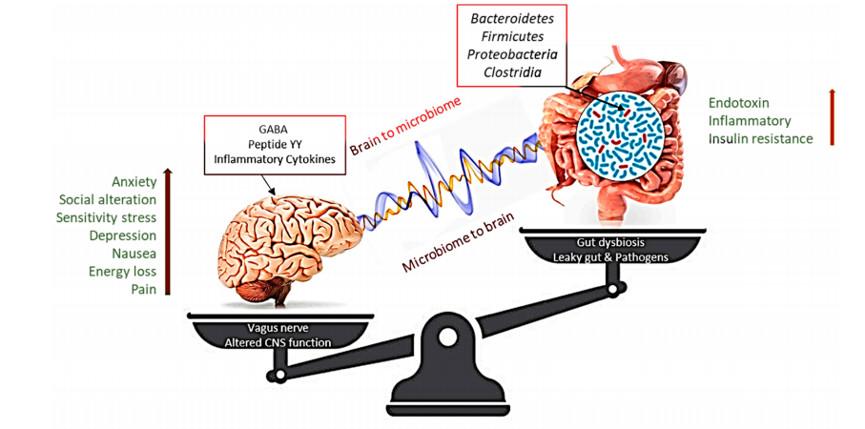
图 2. 微生物组的变化表明,肠道失衡可能会对人体生理产生不利影响,导致肠道脑轴信号不正确以及对中枢神经系统(CNS)功能的相关影响,从而导致肥胖。相反,中枢神经系统水平的压力会影响肠道功能并导致微生物群紊乱。
GABA,即 γ-氨基丁酸。(右箭头)指示内毒素、炎性和胰岛素抵抗的逐渐增加,这与焦虑、社交改变、敏感性应激抑郁、恶心、能量损失和疼痛的增强水平(左箭头)有关。
参考文献:
(滑动下方文字查看)
1. World Health Organization. Overweightand Obesity. Available online:http://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight(accessed on 25 September 2018).
2. Van Baal, P.H.; Hoogenveen, R.T.; deWit, A.G.; Boshuizen, H.C. Estimating health-adjusted life expectancyconditional on risk factors: Results for smoking and obesity. Popul. HealthMetrics 2006, 4, 14.
3. Nascimento Ferreira, M.V.;Rendo-Urteaga, T.; De Moraes, A.C.; Moreno, L.A.; Carvalho, H.B. AbdominalObesity in Children: The Role of Physical Activity, Sedentary Behavior, andSleep Time. In Nutrition in the Prevention and Treatment of Abdominal Obesity;Academic Press: Cambridge, MA, USA, 2019; pp. 81–94. [Google Scholar]
4. FAO; WHO. Guidelines for the Evaluationof Probiotics in Foods. Report of a Joint FAO/WHO Working Group on DraftingGuidelines for the Evaluation of Probiotics in Food; FAO: Rome, Italy; WHO:Geneva, Switzerland, 2002. [Google Scholar]
5. Rajilić-Stojanović, M.; De Vos, W.M. Thefirst 1000 cultured species of the human gastrointestinal microbiota. FEMSMicrobiol. Rev. 2014, 38, 996–1047.
6. Blandino, G.; Inturri, R.; Lazzara, F.;Di Rosa, M.; Malaguarnera, L. Impact of gut microbiota on diabetes mellitus.Diabetes Metab. 2016, 42, 303–315.
7. Gill, H.; Prasad, J. Probiotics,immunomodulation, and health benefits. Adv. Exp. Med. Biol. 2008, 206, 423–454.[Google Scholar]
8. Ashraf, R.; Shah, N.P. Immune SystemStimulation by Probiotic Microorganisms. Crit. Rev. Food Sci. Nutr. 2014, 54,938–956.
9. Kang, J.-H.; Yun, S.-I.; Park, M.-H.;Park, J.-H.; Jeong, S.-Y.; Park, H.-O. Anti-Obesity Effect of Lactobacillusgasseri BNR17 in High-Sucrose Diet-Induced Obese Mice. PLoS ONE 2013, 8,e54617.
10. Park, Y.H.; Kim, J.G.; Shin, Y.W.; Kim,H.S.; Kim, Y.-J.; Chun, T.; Kim, S.H.; Whang, K.Y. Effects of Lactobacillusacidophilus 43121 and a mixture of Lactobacillus casei and Bifidobacteriumlongum on the serum cholesterol level and fecal sterol excretion inhypercholesterolemia-induced pigs. Biosci. Biotechnol. Biochem. 2008, 72,595–600.
11. Sharma, P.; Bhardwaj, P.; Singh, R.Administration of Lactobacillus casei and Bifidobacterium bifidum AmelioratedHyperglycemia, Dyslipidemia, and Oxidative Stress in Diabetic Rats. Int. J.Prev. Med. 2016, 7, 102. [Google Scholar]
12. So, J.-S.; Kwon, H.-K.; Lee, C.-G.; Yi,H.-J.; Park, J.-A.; Lim, S.-Y.; Hwang, K.-C.; Jeon, Y.H.; Im, S.-H.Lactobacillus casei suppresses experimental arthritis by down-regulating Thelper 1 effector functions. Mol. Immunol. 2008, 45, 2690–2699.
13. Larsen, N.; Vogensen, F.K.; Berg,F.W.J.V.D.; Nielsen, D.S.; Andreasen, A.S.; Pedersen, B.K.; Abu Al-Soud, W.;Sørensen, S.J.; Hansen, L.H.; Jakobsen, M. Gut Microbiota in Human Adults withType 2 Diabetes Differs from Non-Diabetic Adults. PLoS ONE 2010, 5, e9085.
14. Kong, Y.; He, M.; McAlister, T.;Seviour, R.; Forster, R. Quantitative Fluorescence In Situ Hybridization ofMicrobial Communities in the Rumens of Cattle Fed Different Diets. Appl.Environ. Microbiol. 2010, 76, 6933–6938.
15. Barrett, E.; Ross, R.; O’Toole, P.;Fitzgerald, G.; Stanton, C. γ-Aminobutyric acid production by culturablebacteria from the human intestine. J. Appl. Microbiol. 2012, 113, 411–417.
16. Khalili, L.; Alipour, B.; Jafar-Abadi,M.A.; Faraji, I.; Hassanalilou, T.; Abbasi, M.M.; Vaghef-Mehrabany, E.; Sani,M.A. The Effects of Lactobacillus casei on Glycemic Response, Serum Sirtuin1and Fetuin-A Levels in Patients with Type 2 Diabetes Mellitus: A RandomizedControlled Trial. Iran. Biomed. J. 2019, 23, 68–77.
17. Sabico, S.; Al-Mashharawi, A.;Al-Daghri, N.M.; Wani, K.; Amer, O.E.; Hussain, D.S.; Ansari, M.G.; Masoud,M.S.; Alokail, M.S.; McTernan, P.G. Effects of a 6-month multi-strain probioticssupplementation in endotoxemic, inflammatory and cardiometabolic status of T2DMpatients: A randomized, double-blind, placebo-controlled trial. Clin. Nutr.2018, 38, 1563–1569.
18. Hu, C.; Wong, F.S.; Wen, L. Type 1diabetes and gut microbiota: Friend or foe? Pharmacol. Res. 2015, 98, 9–15.
19. Ljungberg, M.; Korpela, R.; Ilonen, J.;Ludvigsson, J.; Vaarala, O. Probiotics for the Prevention of Beta CellAutoimmunity in Children at Genetic Risk of Type 1 Diabetes—The PRODIA Study.Ann. N. Y. Acad. Sci. 2006, 1079, 360–364.
20. Hartstra, A.V.; Bouter, K.E.; Bäckhed,F.; Nieuwdorp, M. Insights into the role of the microbiome in obesity and type2 diabetes. Diabetes Care 2015, 38, 159–165.
21. Grover, S.; Rashmi, H.M.; Srivastava,A.K.; Batish, V.K. Probiotics for human health—New innovations and emergingtrends. Gut Pathog. 2012, 4, 15.
22. Szulinska, M.; Łoniewski, I.; VanHemert, S.; Sobieska, M.; Bogdański, P. Dose-Dependent Effects of MultispeciesProbiotic Supplementation on the Lipopolysaccharide (LPS) Level andCardiometabolic Profile in Obese Postmenopausal Women: A 12-Week RandomizedClinical Trial. Nutrients 2018, 10, 773.
23. Chan, P.A.; Robinette, A.; Montgomery,M.; Almonte, A.; Cu-Uvin, S.; Lonks, J.R.; Chapin, K.C.; Kojic, E.M.; Hardy,E.J. Extragenital Infections Caused by Chlamydia trachomatis and Neisseriagonorrhoeae: A Review of the Literature. Infect. Dis. Obstet. Gynecol. 2016,2016, 1–17.
24. Le Barz, M.; Anhê, F.F.; Varin, T.V.;Desjardins, Y.; Levy, E.; Roy, D.; Urdaci, M.C.; Marette, A. Probiotics asComplementary Treatment for Metabolic Disorders. Diabetes Metab. J. 2015, 39,291–303.
25. International Diabetes Federation. IDFDiabetes Atlas; IDF: Watermael-Boitsfort, Belgium, 2017. Available online:http://www.diabetesatlas.org/resources/2017-atlas.html (accessed on 12 July2019).
26. Kobyliak, N.; Conte, C.; Cammarota, G.;Haley, A.P.; Štyriak, I.; Gaspar, L.; Fusek, J.; Rodrigo, L.; Kruzliak, P.Probiotics in prevention and treatment of obesity: A critical view. Nutr. Metab.2016, 13, 14.
27. Barrett, H.L.; Callaway, L.K.; Nitert,M.D. Probiotics: A potential role in the prevention of gestational diabetes?Acta Diabetol. 2012, 49, 1–13.
28. Mishra, A.K.; Dubey, V.; Ghosh, A.R.;Information, P.E.K.F.C. Obesity: An overview of possible role(s) of guthormones, lipid sensing and gut microbiota. Metablism 2016, 65, 48–65.
29. Zhao, X.; Higashikawa, F.; Noda, M.;Kawamura, Y.; Matoba, Y.; Kumagai, T.; Sugiyama, M. The Obesity and Fatty LiverAre Reduced by Plant-Derived Pediococcus pentosaceus LP28 in High FatDiet-Induced Obese Mice. PLoS ONE 2012, 7, e30696.
30. Park, S.-Y.; Cho, S.-A.; Lee, M.-K.;Lim, S.-D. Effect of Lactobacillus plantarum FH185 on the Reduction ofAdipocyte Size and Gut Microbial Changes in Mice with Diet-induced Obesity.Food Sci. Anim. Resour. 2015, 35, 171–178.
31. Park, S.; Ji, Y.; Jung, H.Y.; Park, H.;Kang, J.; Choi, S.H.; Shin, H.; Hyun, C.K.; Kim, K.T.; Holzapfel, W.H.Lactobacillus plantarum HAC01 regulates gut microbiota and adipose tissueaccumulation in a diet-induced obesity murine model. Appl. Microbiol.Biotechnol. 2017, 101, 1605–1614.
32. Wu, C.-C.; Weng, W.-L.; Lai, W.-L.;Tsai, H.-P.; Liu, W.-H.; Lee, M.-H.; Tsai, Y.-C. Effect of Lactobacillusplantarum Strain K21 on High-Fat Diet-Fed Obese Mice. Evid. Based Complement.Altern. Med. 2015, 2015, 1–9. [Google Scholar]
33. Park, J.E.; Oh, S.H.; Cha, Y.S.Lactobacillus plantarum LG42 isolated from gajami sik-hae decreases body andfat pad weights in diet-induced obese mice. J. Appl. Microbiol. 2014, 116,145–156.
34. Callaway, L.K.; McIntyre, H.D.;Barrett, H.L.; Foxcroft, K.; Tremellen, A.; Lingwood, B.E.; Tobin, J.M.;Wilkinson, S.; Kothari, A.; Morrison, M.; et al. Probiotics for the Preventionof Gestational Diabetes Mellitus in Overweight and Obese Women: Findings Fromthe SPRING Double-blind Randomized Controlled Trial. Diabetes Care 2019, 42,dc182248.
35. Okesene-Gafa, K.A.M.; Li, M.; McKinlay,C.J.D.; Taylor, R.S.; Rush, E.C.; Wall, C.R.; Wilson, J.; Murphy, R.; Taylor,R.; Thompson, J.M.D.; et al. Effect of antenatal dietary interventions inmaternal obesity on pregnancy weight-gain and birthweight: Healthy Mums andBabies (HUMBA) randomized trial. Am. J. Obstet. Gynecol. 2019, 221, 1–13.
36. Krumbeck, J.A.; Rasmussen, H.E.;Hutkins, R.W.; Clarke, J.; Shawron, K.; Keshavarzian, A.; Walter, J. ProbioticBifidobacterium strains and galacto oligosaccharides improve intestinal barrierfunction in obese adults but show no synergism when used together assynbiotics. Microbiome 2018, 6, 121.
37. Kim, J.; Yun, J.M.; Kim, M.K.; Kwon,O.; Cho, B. Lactobacillus gasseri BNR17 Supplementation Reduces the VisceralFat Accumulation and Waist Circumference in Obese Adults: A Randomized,Double-Blind, Placebo-Controlled Trial. J. Med. Food 2018, 21, 454–461.
38. Minami, J.; Iwabuchi, N.; Tanaka, M.;Yamauchi, K.; Xiao, J.-Z.; Abe, F.; Sakane, N. Effects of Bifidobacterium breveB-3 on body fat reductions in pre-obese adults: A randomized, double-blind,placebo-controlled trial. Biosci. Microbiota Food Health 2018, 37, 67–75.
39. Ogawa, A.; Kadooka, Y.; Kato, K.;Shirouchi, B.; Sato, M. Lactobacillus gasseri SBT2055 reduces postprandial andfasting serum non-esterified fatty acid levels in Japanesehypertriacylglycerolemic subjects. Lipids Health Dis. 2014, 13, 36.
40. Dietrich, C.G.; Kottmann, T.; Alavi, M.Commercially available probiotic drinks containing Lactobacillus caseiDN-114001 reduce antibiotic-associated diarrhea. World J. Gastroenterol. 2014,20, 15837–15844.
41. Iqbal, M.Z.; Qadir, M.I.; Hussain, T.;Janbaz, K.H.; Khan, Y.H.; Ahmad, B. Review: Probiotics and their beneficialeffects against various diseases. Pak. J. Pharm. Sci. 2014, 27, 405–415.[Google Scholar]
42. Slavin, J. Fiber and Prebiotics:Mechanisms and Health Benefits. Nutrients 2013, 5, 1417–1435.
43. Bejar, W.; Hamden, K.; Ben Salah, R.;Chouayekh, H. Lactobacillus plantarum TN627 significantly reduces complicationsof alloxan-induced diabetes in rats. Anaerobe 2013, 24, 4–11.
44. Sakai, T.; Taki, T.; Nakamoto, A.;Shuto, E.; Tsutsumi, R.; Toshimitsu, T.; Makino, S.; Ikegami, S. Lactobacillusplantarum OLL2712 regulates glucose metabolism in C57BL/6 mice fed a high-fatdiet. J. Nutr. Sci. Vitaminol. 2013, 59, 144–147.
45. Yakovlieva, M.; Tacheva, T.; Mihaylova,S.; Tropcheva, R.; Trifonova, K.; Tolesmall ka, C.A.; Danova, S.; Vlaykova, T.Influence of Lactobacillus brevis 15 and Lactobacillus plantarum 13 on bloodglucose and body weight in rats after high-fructose diet. Benef. Microbes 2015,6, 505–512.
46. Huang, H.-Y.; Korivi, M.; Tsai, C.-H.;Yang, J.-H.; Tsai, Y.-C. Supplementation of Lactobacillus plantarum K68 andFruit-Vegetable Ferment along with High Fat-Fructose Diet Attenuates MetabolicSyndrome in Rats with Insulin Resistance. Evid. Based Complement. Altern. Med.2013, 2013, 1–12. [Google Scholar]
47. Li, X.; Yin, B.; Fang, D.; Jiang, T.;Zhao, J.; Wang, N.; Fang, S.; Zhang, H.; Wang, G.; Chen, W. Effects ofLactobacillus plantarum CCFM0236 on hyperglycaemia and insulin resistance inhigh-fat and streptozotocin-induced type 2 diabetic mice. J. Appl. Microbiol.2016, 121, 1727–1736.
48. Zuo, T.; Ng, S.C. The Gut Microbiota inthe Pathogenesis and Therapeutics of Inflammatory Bowel Disease. Front.Microbiol. 2018, 9, 2247.
49. Opazo, M.C.; Ortega-Rocha, E.M.;Coronado-Arrázola, I.; Bonifaz, L.C.; Boudin, H.; Neunlist, M.; Bueno, S.M.;Kalergis, A.M.; Riedel, C.A. Intestinal microbiota influences non-intestinalrelated autoimmune diseases. Front. Microbiol. 2018, 9, 432.
50. Jumpertz, R.; Le, D.S.; Turnbaugh,P.J.; Trinidad, C.; Bogardus, C.; Gordon, J.I.; Krakoff, J. Energy-balancestudies reveal associations between gut microbes, caloric load, and nutrientabsorption in humans. Am. J. Clin. Nutr. 2011, 94, 58–65.
51. Ignacio, A.; Fernandes, M.; Rodrigues,V.; Groppo, F.; Cardoso, A.; Avila-Campos, M.; Nakano, V.; Avila-Campos, M.Correlation between body mass index and faecal microbiota from children. Clin.Microbiol. Infect. 2016, 22, 1–8.
52. Patil, D.P.; Dhotre, D.P.; Chavan,S.G.; Sultan, A.; Jain, D.S.; Lanjekar, V.B.; Gangawani, J.; Shah, P.S.;Todkar, J.S.; Shah, S.; et al. Molecular analysis of gut microbiota in obesityamong Indian individuals. J. Biosci. 2012, 37, 647–657.
53. Zhang, H.; DiBaise, J.K.; Zuccolo, A.;Kudrna, D.; Braidotti, M.; Yu, Y.; Parameswaran, P.; Crowell, M.D.; Wing, R.;Rittmann, B.E.; et al. Human gut microbiota in obesity and after gastricbypass. Proc. Natl. Acad. Sci. USA 2009, 106, 2365–2370.
54. Santacruz, A.; Collado, M.C.;García-Valdés, L.; Segura, M.T.; Martín-Lagos, J.A.; Anjos, T.; Martí-Romero,M.; Lopez, R.M.; Florido, J.; Campoy, C.; et al. Gut microbiota composition isassociated with body weight, weight gain and biochemical parameters in pregnantwomen. Br. J. Nutr. 2010, 104, 83–92.
55. Allen, J.M.; Jaggers, R.M.; Solden,L.M.; Loman, B.R.; Davies, R.H.; Mackos, A.R.; Ladaika, C.A.; Berg, B.M.;Chichlowski, M.; Bailey, M.T. Dietary oligosaccharides attenuate stress-induceddisruptions in immune reactivity and microbial B-vitamin metabolism. FrontImmunol. 2019, 10, 1774.
56. Fei, N.; Zhao, L. An opportunisticpathogen isolated from the gut of an obese human causes obesity in germfreemice. ISME J. 2013, 7, 880–884.
57. Bervoets, L.; Van Hoorenbeeck, K.;Kortleven, I.; Van Noten, C.; Hens, N.; Vael, C.; Goossens, H.; Desager, K.N.;Vankerckhoven, V. Differences in gut microbiota composition between obese andlean children: A cross-sectional study. Gut Pathog. 2013, 5, 10.
58. Duncan, S.H.; Belenguer, A.; Holtrop,G.; Johnstone, A.M.; Flint, H.J.; Lobley, G.E. Reduced dietary intake ofcarbohydrates by obese subjects results in decreased concentrations of butyrateand butyrate-producing bacteria in feces. Appl. Environ. Microbiol. 2007, 73,1073–1078.
59. Costa, F.R.; Françozo, M.C.; DeOliveira, G.G.; Ignacio, A.; Castoldi, A.; Zamboni, D.S.; Ramos, S.G.; Câmara,N.O.; De Zoete, M.R.; Palm, N.W.; et al. Gut microbiota translocation to thepancreatic lymph nodes triggers NOD2 activation and contributes to T1D onset.J. Exp. Med. 2016, 213, 1223–1239.
60. Murugesan, S.; Ulloa-Martínez, M.;Martinez-Rojano, H.; Galván-Rodríguez, F.M.; Miranda-Brito, C.; Romano, M.C.;Piña-Escobedo, A.; Pizano-Zárate, M.L.; Hoyo-Vadillo, C.; García-Mena, J. Studyof the diversity and short-chain fatty acids production by the bacterialcommunity in overweight and obese Mexican children. Eur. J. Clin. Microbiol.Infect. Dis. 2015, 34, 1337–1346.
61. Verdam, F.J.; Fuentes, S.; De Jonge,C.; Zoetendal, E.G.; Erbil, R.; Greve, J.W.; Buurman, W.A.; De Vos, W.M.;Rensen, S.S. Human intestinal microbiota composition is associated with localand systemic inflammation in obesity. Obesity 2013, 21, E607–E615.
62. Kasai, C.; Sugimoto, K.; Moritani, I.;Tanaka, J.; Oya, Y.; Inoue, H.; Tameda, M.; Shiraki, K.; Ito, M.; Takei, Y.; etal. Comparison of the gut microbiota composition between obese and non-obeseindividuals in a Japanese population, as analyzed by terminal restrictionfragment length polymorphism and next-generation sequencing. BMC Gastroenterol.2015, 15, 100.
63. Payahoo, L.; Khajebishak, Y.;Ostadrahimi, A. Akkermansia muciniphila bacteria: A new perspective on themanagement of obesity: An updated review. Rev. Med. Microbiol. 2019, 30, 83–89.
64. Million, M.; Angelakis, E.; Paul, M.;Armougom, F.; Leibovici, L.; Raoult, D. Comparative meta-analysis of the effectof Lactobacillus species on weight gain in humans and animals. Microb. Pathog.2012, 53, 100–108.
65. Qiao, Y.; Sun, J.; Xia, S.; Li, L.; Li,Y.; Wang, P.; Shi, Y.; Le, G. Effects of different Lactobacillus reuteri oninflammatory and fat storage in high-fat diet-induced obesity mice model. J.Funct. Foods 2015, 14, 424–434.
66. Ridlon, J.M.; Kang, D.J. Hylemon PB.Bile salt biotransformations by human intestinal bacteria. J. Lipid Res. 2006,47, 241–259.
67. Binder, H.J.; Filburn, B.; Floch, M.Bile acid inhibition of intestinal anaerobic organisms. Am. J. Clin. Nutr.1975, 28, 119–125.
68. Kurdi, P.; Kawanishi, K.; Mizutani, K.;Yokota, A. Mechanism of Growth Inhibition by Free Bile Acids in Lactobacilliand Bifidobacteria. J. Bacteriol. 2006, 188, 1979–1986.
69. Fiorucci, S.; Mencarelli, A.;Palladino, G.; Cipriani, S. Bile-acid-activated receptors: Targeting TGR5 andfarnesoid-X-receptor in lipid and glucose disorders. Trends Pharmacol. Sci.2009, 30, 570–580.
70. Thomas, C.; Gioiello, A.; Noriega, L.;Strehle, A.; Oury, J.; Rizzo, G.; Macchiarulo, A.; Yamamoto, H.; Mataki, C.;Pruzanski, M.; et al. TGR5-mediated bile acid sensing controls glucosehomeostasis. Cell Metab. 2009, 10, 167–177.
71. McGavigan, A.K.; Garibay, D.; Henseler,Z.M.; Chen, J.; Bettaieb, A.; Haj, F.G.; Ley, R.E.; Chouinard, M.L.; Cummings,B.P. TGR5 contributes to glucoregulatory improvements after vertical sleevegastrectomy in mice. Gut 2017, 66, 226–234.
72. Parséus, A.; Sommer, N.; Sommer, F.;Caesar, R.; Molinaro, A.; Ståhlman, M.; Greiner, T.U.; Perkins, R.; Bäckhed, F.Microbiota-induced obesity requires farnesoid X receptor. Gut 2017, 66,429–437.
73. Flint, H.J.; Bayer, E.A.; Rincon, M.T.;Lamed, R.; White, B.A. Polysaccharide utilization by gut bacteria: Potentialfor new insights from genomic analysis. Nat. Rev. Genet. 2008, 6, 121–131.
74. Riley, L.W.; Raphael, E.; Faerstein, E.Obesity in the United States—Dysbiosis from Exposure to Low-Dose Antibiotics?Front. Public Health 2013, 1, 69.
75. Canfora, E.E.; Meex, R.C.; Venema, K.;Blaak, E.E. Gut microbial metabolites in obesity, NAFLD and T2DM. Nat. Rev.Endocrinol. 2019, 1.
76. Brinkworth, G.D.; Noakes, M.; Clifton,P.M.; Bird, A.R. Comparative effects of very low-carbohydrate, high-fat andhigh-carbohydrate, low-fat weight-loss diets on bowel habit and faecalshort-chain fatty acids and bacterial populations. Br. J. Nutr. 2009, 101,1493.
77. Brown, A.J.; Goldsworthy, S.M.; Barnes,A.A.; Eilert, M.M.; Tcheang, L.; Daniels, D.; Muir, A.I.; Wigglesworth, M.J.;Kinghorn, I.; Fraser, N.J.; et al. The Orphan G protein-coupled receptors GPR41and GPR43 are activated by propionate and other short chain carboxylic acids.J. Biol. Chem. 2003, 278, 11312–11319.
78. Xu, H.; Barnes, G.T.; Yang, Q.; Tan,G.; Yang, D.; Chou, C.J.; Sole, J.; Nichols, A.; Ross, J.S.; Tartaglia, L.A.;et al. Chronic inflammation in fat plays a crucial role in the development ofobesity-related insulin resistance. J. Clin. Investig. 2003, 112, 1821–1830.
79. Cani, P.D.; Amar, J.; Iglesias, M.A.;Poggi, M.; Knauf, C.; Bastelica, D.; Neyrinck, A.M.; Fava, F.; Tuohy, K.M.;Chabo, C.; et al. Metabolic Endotoxemia Initiates Obesity and InsulinResistance. Diabetes 2007, 56, 1761–1772.
80. Dalby, M.J.; Aviello, G.; Ross, A.W.;Walker, A.W.; Barrett, P.; Morgan, P.J. Diet induced obesity is independent ofmetabolic endotoxemia and TLR4 signalling, but markedly increases hypothalamicexpression of the acute phase protein, SerpinA3N. Sci. Rep. 2018, 8, 15648.
81. Amar, J.; Burcelin, R.; Ruidavets,J.B.; Cani, P.D.; Fauvel, J.; Alessi, M.C.; Chamontin, B.; Ferriéres, J. Energyintake is associated with endotoxemia in apparently healthy men. Am. J. Clin.Nutr. 2008, 87, 1219–1223.
82. Hotamisligil, G.S. Inflammation andmetabolic disorders. Nature 2006, 444, 860–867.
83. Chawla, A. Nuclear Receptors and LipidPhysiology: Opening the X-Files. Science 2001, 294, 1866–1870.
84. Glass, C.K.; Ogawa, S. Combinatorial rolesof nuclear receptors in inflammation and immunity. Nat. Rev. Immunol. 2006, 6,44–55.
85. Wellen, K.E.; Hotamisligil, G.S.Inflammation, stress, and diabetes. J. Clin. Investig. 2005, 115,1111–1119.
86. Ge, H.; Li, X.; Weiszmann, J.; Wang,P.; Baribault, H.; Chen, J.-L.; Tian, H.; Li, Y. Activation of GProtein-Coupled Receptor 43 in Adipocytes Leads to Inhibition of Lipolysis andSuppression of Plasma Free Fatty Acids. Endocrinology 2008, 149,4519–4526.
87. Semenkovich, C.F. Insulin resistanceand atherosclerosis. J. Clin. Investig. 2006, 116, 1813–1822.
88. Bäckhed, F.; Ding, H.; Wang, T.;Hooper, L.V.; Koh, G.Y.; Nagy, A.; Semenkovich, C.F.; Gordon, J.I. The gutmicrobiota as an environmental factor that regulates fat storage. Proc. Natl.Acad. Sci. USA 2004, 101, 15718–15723.
89. Shapiro, H.; Suez, J.; Elinav, E.Personalized microbiome-based approaches to metabolic syndrome management andprevention. J. Diabetes 2017, 9, 226–236.
90. Wang, Z.; Klipfell, E.; Bennett, B.J.;Koeth, R.; Levison, B.S.; Dugar, B.; Feldstein, A.E.; Britt, E.B.; Fu, X.;Chung, Y.-M.; et al. Gut flora metabolism of phosphatidylcholine promotescardiovascular disease. Nature 2011, 472, 57–63.
91. Koeth, R.A.; Wang, Z.; Levison, B.S.;Buffa, J.A.; Org, E.; Sheehy, B.T.; Britt, E.B.; Fu, X.; Wu, Y.; Li, L.; et al.Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat,promotes atherosclerosis. Nat. Med. 2013, 19, 576–585.
92. Chiang, J.Y.L. Bile acids: Regulationof synthesis. J. Lipid Res. 2009, 50, 1955–1966.
93. Vrieze, A.; Out, C.; Fuentes, S.;Jonker, L.; Reuling, I.; Kootte, R.S.; Van Nood, E.; Holleman, F.; Knaapen, M.;Romijn, J.A.; et al. Impact of oral vancomycin on gut microbiota, bile acidmetabolism, and insulin sensitivity. J. Hepatol. 2014, 60, 824–831.
94. Reijnders, D.; Goossens, G.H.; Hermes,G.D.; Neis, E.P.; Van Der Beek, C.M.; Most, J.; Holst, J.J.; Lenaerts, K.;Kootte, R.S.; Nieuwdorp, M.; et al. Effects of Gut Microbiota Manipulation byAntibiotics on Host Metabolism in Obese Humans: A Randomized Double-BlindPlacebo-Controlled Trial. Cell Metab. 2016, 24, 341.
95. Delgado, T.C. Glutamate and GABA inAppetite Regulation. Front. Endocrinol. 2013, 4, 103.
96. Duca, F.A.; Swartz, T.D.; Sakar, Y.;Covasa, M. Increased Oral Detection, but Decreased Intestinal Signaling forFats in Mice Lacking Gut Microbiota. PLoS ONE 2012, 7, e39748.
97. Ley, R.E.; Bäckhed, F.; Turnbaugh, P.;Lozupone, C.A.; Knight, R.D.; Gordon, J.I. Obesity alters gut microbialecology. Proc. Natl. Acad. Sci. USA 2005, 102, 11070–11075.
98. Turnbaugh, P.J.; Gordon, J.I. The coregut microbiome, energy balance and obesity. J. Physiol. 2009, 587, 4153–4158.
99. Ussar, S.; Griffin, N.W.; Bézy, O.;Fujisaka, S.; Vienberg, S.; Softic, S.; Deng, L.; Bry, L.; Gordon, J.I.; Kahn,C.R. Interactions between Gut Microbiota, Host Genetics and Diet Modulate thePredisposition to Obesity and Metabolic Syndrome. Cell Metab. 2015, 22,516–530.
100. Goodrich, J.K.; Waters, J.L.; Poole,A.C.; Sutter, J.L.; Koren, O.; Blekhman, R.; Beaumont, M.; Van Treuren, W.;Knight, R.; Bell, J.T.; et al. Human Genetics Shape the Gut Microbiome. Cell2014, 159, 789–799.
101. Liu, R.; Hong, J.; Xu, X.; Feng, Q.;Zhang, D.; Gu, Y.; Shi, J.; Zhao, S.; Liu, W.; Wang, X.; et al. Gut microbiomeand serum metabolome alterations in obesity and after weight-loss intervention.Nat. Med. 2017, 23, 859–868.
102. Bodenlos, J.S.; Schneider, K.L.;Oleski, J.; Gordon, K.; Rothschild, A.J.; Pagoto, S.L. Vagus nerve stimulationand food intake: Effect of body mass index. J. Diabetes Sci. Technol. 2014, 8,590–595.
103. Meng, F.; Han, Y.; Srisai, D.;Belakhov, V.; Farias, M.; Xu, Y.; Palmiter, R.D.; Baasov, T.; Wu, Q. Newinducible genetic method reveals critical roles of GABA in the control offeeding and metabolism. Proc. Natl. Acad. Sci. USA 2016, 113, 3645–3650.
104. Schellekens, H.; Dinan, T.G.; Cryan,J.F. Lean mean fat reducing “ghrelin” machine: Hypothalamic ghrelin and ghrelinreceptors as therapeutic targets in obesity. Neuropharmacology 2010, 58, 2–16.
105. Heisler, L.K.; Jobst, E.E.; Sutton,G.M.; Zhou, L.; Borok, E.; Thornton-Jones, Z.; Liu, H.Y.; Zigman, J.M.;Balthasar, N.; Kishi, T.; et al. Serotonin Reciprocally Regulates MelanocortinNeurons to Modulate Food Intake. Neuron 2006, 51, 239–249.
106. Xu, Y.; Jones, J.E.; Kohno, D.;Williams, K.W.; Lee, C.E.; Choi, M.J.; Anderson, J.G.; Heisler, L.K.; Zigman,J.M.; Lowell, B.B.; et al. 5-HT2CRs expressed by pro-opiomelanocortin neuronsregulate energy homeostasis. Neuron 2008, 60, 582–589.
107. Sandhu, K.V.; Sherwin, E.;Schellekens, H.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Feeding themicrobiota-gut-brain axis: Diet, microbiome, and neuropsychiatry. Transl. Res.2016, 179, 223–244.
108. Everard, A.; Cani, P.D. Gut microbiotaand GLP-1. Rev. Endocr. Metab. Disord. 2014, 15, 189–196.
109. Niwa, T.; Takeda, N.; Tatematsu, A.;Maeda, K. Accumulation of indoxyl sulfate, an inhibitor of drug-binding, inuremic serum as demonstrated by internal-surface reversed-phase liquidchromatography. Clin. Chem. 1988, 34, 2264–2267.
110. Frost, G.; Sleeth, M.L.; Sahuri-Arisoylu,M.; Lizarbe, B.; Cerdan, S.; Brody, L.; Anastasovska, J.; Ghourab, S.; Hankir,M.; Zhang, S.; et al. The short-chain fatty acid acetate reduces appetite via acentral homeostatic mechanism. Nat. Commun. 2014, 5, 3611.
111. Perry, R.J.; Peng, L.; Barry, N.A.Acetate mediates a microbiome-brain-beta-cell axis to promote metabolicsyndrome. Nature 2016, 534, 213–217.
112. Swartz, T.D.; Duca, F.A.; de Wouters,T.; Sakar, Y.; Covasa, M. Up-regulation of intestinal type 1 taste receptor 3and sodium glucose luminal transporter-1 expression and increased sucroseintake in mice lacking gut microbiota. Br. J. Nutr. 2012, 107, 621–630.
原文链接:https://www.mdpi.com/2076-2607/7/10/456/htm#B66-microorganisms-07-00456
作者|Kaliyan Barathikannan 等人
翻译|gemiu
审校|617